 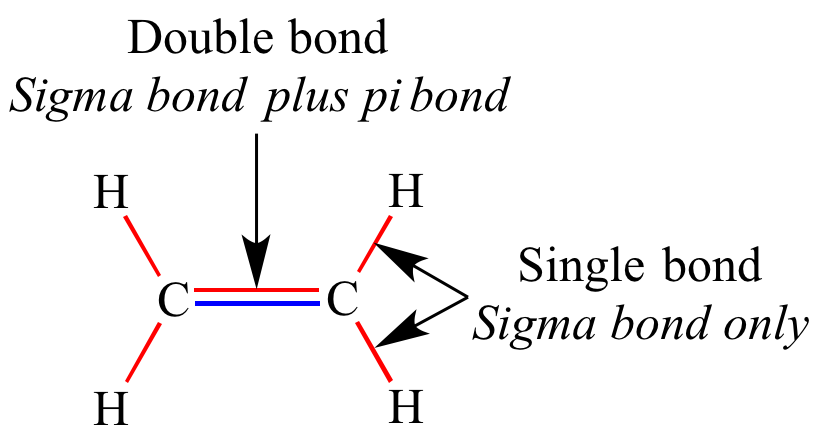
The distribution is valid for atoms or molecules constituting ideal gases. Each of these atomic orbitals has an electron density of zero at a shared nodal plane that passes through the two bonded nuclei. 
In complex analysis, is used in the Weierstrass sigma-function. In chemistry, pi bonds ( bonds) are covalent chemical bonds, in each of which two lobes of an orbital on one atom overlap with two lobes of an orbital on another atom, and in which this overlap occurs laterally. In applied mathematics, (T) denotes the spectrum of a linear map T. Β = 1 / k B T, for reversible processes onlyīelow are useful results from the Maxwell–Boltzmann distribution for an ideal gas, and the implications of the Entropy quantity. finite measure (see measure theory) In number theory, is included in various divisor functions, especially the sigma function or sum-of-divisors function. General derived quantities Quantity (Common Name/s) General basic quantities Quantity (Common Name/s) Thus the pi molecular orbital is higher in energy and is the highest occupied molecular orbital (the HOMO). 
The pi bond between the two carbon atoms has one node in the plane of the molecule. Many of the definitions below are also used in the thermodynamics of chemical reactions. The sigma bond between the two carbon atoms does not have a node in the plane of the molecule. Just as there is a ((2l+1)) degeneracy in the spherical wavefunctions, there is also an important degeneracy pattern in the wavefunctions of diatomic molecules. Main articles: List of thermodynamic properties, Thermodynamic potential, Free entropy, and Defining equation (physical chemistry) The total number of electrons around each individual atom consists of six nonbonding electrons and two shared (i.e. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
